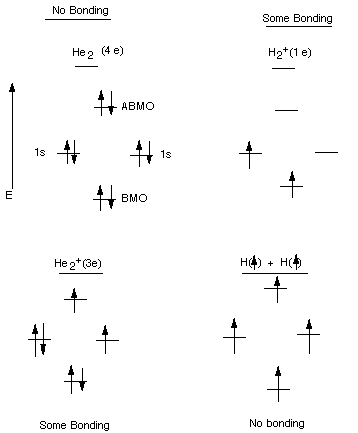
The last (and only) level of helium's electronic configuration is 1s2, and therefore He has 2 valence electrons. This means it has two electrons in s orbitals with a principal quantum number of 1. In the periodic table, the elements are listed in order of increasing atomic number Z. Helium has two electrons in total, and according to the aufbau principle, it adopts the electronic configuration 1s2. The number of electrons in each element’s electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. Subsequently, one may also ask, what is the valence electrons for helium? Helium has a complete shell of electrons, and in this form the atom does not readily accept any extra electrons or join with anything to make covalent compounds. Now I am going to show you how many valence electrons helium have in just 5 steps. 
Helium is one of the most unreactive elements, so it was commonly believed that helium compounds do not exist at all. Secondly, does helium have any compounds? Helium compounds. 
The peripheral shell of a molecule can accomodate a maximum of 8 electrons. Below are their quantum numbers (N - energy, L - angular momentum, M - magnetic moment, S - spin ). Furthermore, unexpected compounds, such as. The number of valence electrons in a Helium atom - 2. Valence electrons are the electrons located in an atom's outermost shell. an insulator due to strong core-core orbital overlap leading to interstitial valence electron localization13. For H, He, Li, Be, and B (Z from 1 to 5) this is not correct, as H and He have only the n 1 shell occupied, which fills at 2 electrons, and the other three prefer to give up their n 2 shell electr. Therefore, neutral helium will only have 2 electrons surrounding its nucleus. Answer (1 of 4): To answer this, let’s look at what valence electrons are: Valence electrons - Electrons in the outer shell of an atom that are involved in reactions (by bonding). Answer (1 of 4): Your statement is an over-generalization that is true only for some elements. Helium is located in period 1 and has an atomic number equal to 2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
